What causes CIDP?
To understand what causes CIDP, you need to understand the relationship between neurons and myelin:
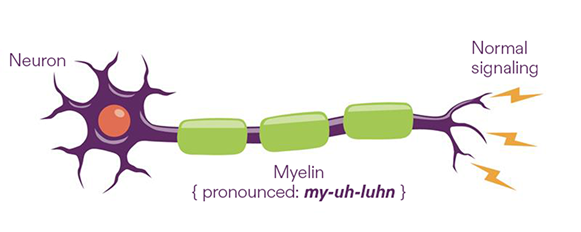
Myelin protects neurons
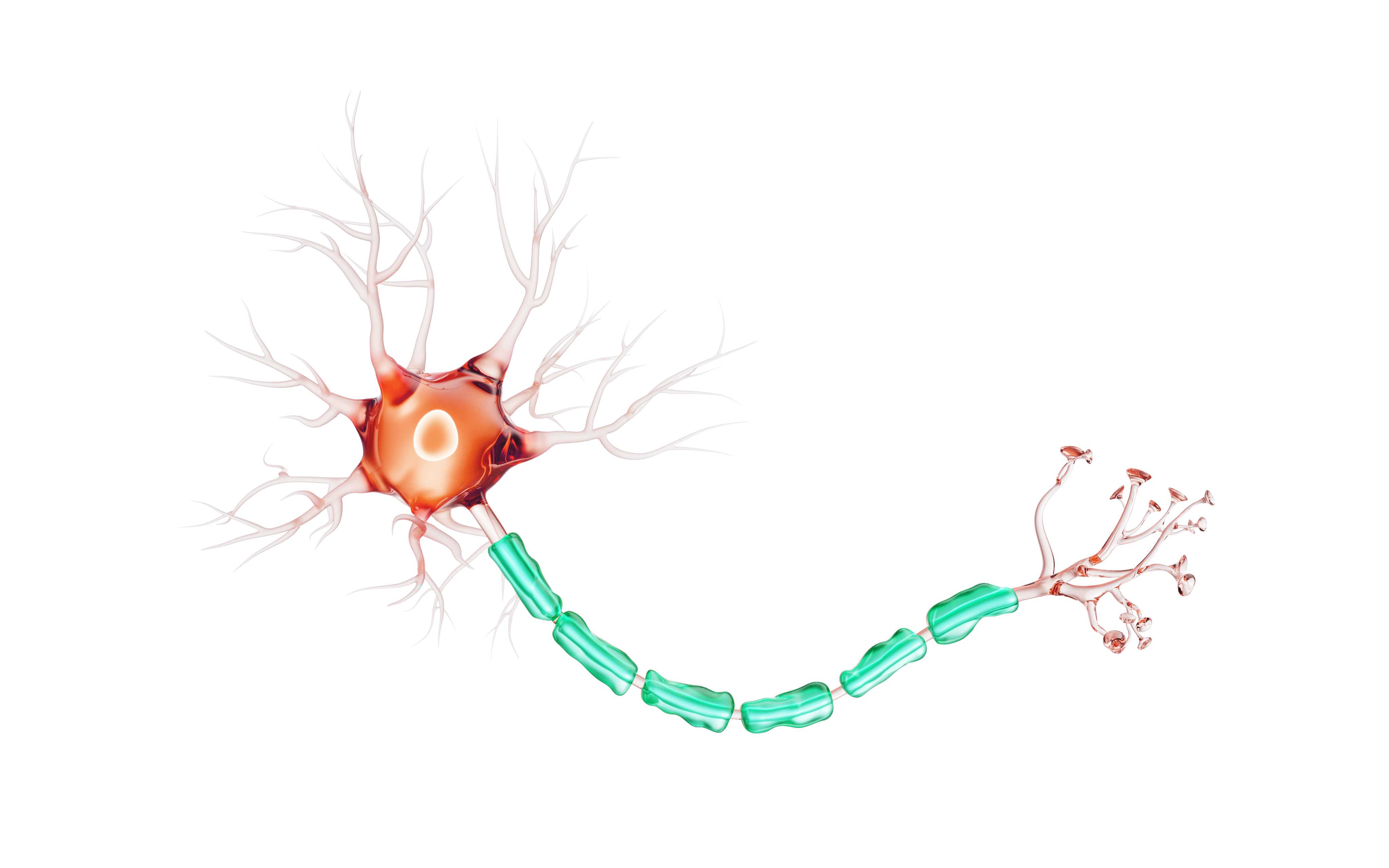
Nerves are made up of neurons, which deliver messages between the brain and the rest of the body to perform actions like picking up a cup or taking a step.
Myelin is a protective layer around a neuron—like insulation around a wire—that helps messages get to their destinations.
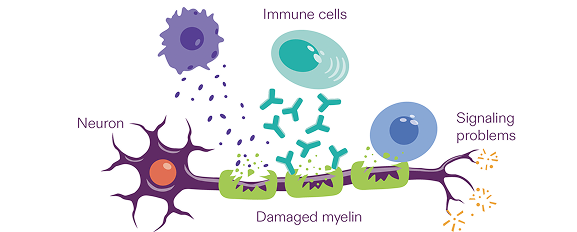
CIDP attacks myelin
The immune system is designed to spot foreign germs in the body and fight them off.
But with CIDP, the immune system thinks the myelin in your arms or legs is foreign and attacks it. This causes the messages neurons deliver to be slowed or lost.
Symptoms of CIDP
The most common symptom experienced with CIDP is weakness in the arms and legs. Some people may also feel a prickling sensation and numbness. Symptoms can happen in waves, coming and going over time, or worsen gradually.
Symptoms of CIDP may include:
Weakness in arms and legs
Loss of coordination and difficulty with walking or standing up
Difficulty with fine motor skills
Loss of reflexes
Prickling sensation and numbness
Pain and fatigue

Your healthcare team
Get results with PANZYGA
Learn about the clinical study, where a majority of patients saw improvement with PANZYGA
Getting started with PANZYGA
View helpful info on what to expect before, during and after PANZYGA infusions
Find savings and resources
View co-pay program info, where eligible patients may pay as little as $0,* and explore other helpful materials and tools
*Eligible, commercially insured patients may receive a maximum benefit of $12,500 per year or the cost of a patient’s co-pay in a 12-month period (whichever is less) for claims received by the program. No membership fees are required. Federal and state healthcare beneficiaries are not eligible. The PANZYGA Co-Pay Program is good only in the US and Puerto Rico. Terms and conditions/eligibility requirements apply. See full Terms and Conditions on the Financial assistance programs page.
Want to connect with other patients diagnosed with CIDP?
The GBS|CIDP Foundation offers education and support to individuals and families affected by CIDP